Liquid Chlorine
1) Description
Name: ........................................................................................... Chlorine
Chemical Formula: ...................................................................Cl2
CAS No: ........................................................................................7782-50-5
Other Names: ............................................................................. Molecular chlorine
Uses: ..............................................................................................Industrial, Domestic
Physical state: .............................................................................Liquid, Gas, Liquefied Gas
Color: .............................................................................................Yellow-Green
Odor: ..............................................................................................Irritating, bleach-like
Boiling Point: ...............................................................................-34 ℃ -29 ℉
Freezing Point: ..............................................................................-101.5℃ (-150℉)
Flammability (solid, gas): .........................................................N/A
Vapor pressure: ...........................................................................6.8 atm
Viscosity (20℃ (liquid)): ...........................................................0.134 mPa.sec at 20℃ (gas); 0.346 mPa.sec
2) Hazards
Chlorine is considered Immediately Dangerous to Life or Health (IDLH). NIOSH (1994).
H270: May cause or intensify fire; oxidizer.
H315: Causes skin irritation.
H319: Causes serious eye irritation
H331: Toxic if inhaled
H335: May cause respiratory irritation
H400: Very toxic to aquatic life
Reactivity: Strong Oxidizing Agent, Water-Reactive, incompatible with Alcohols. Reacts with most metals.
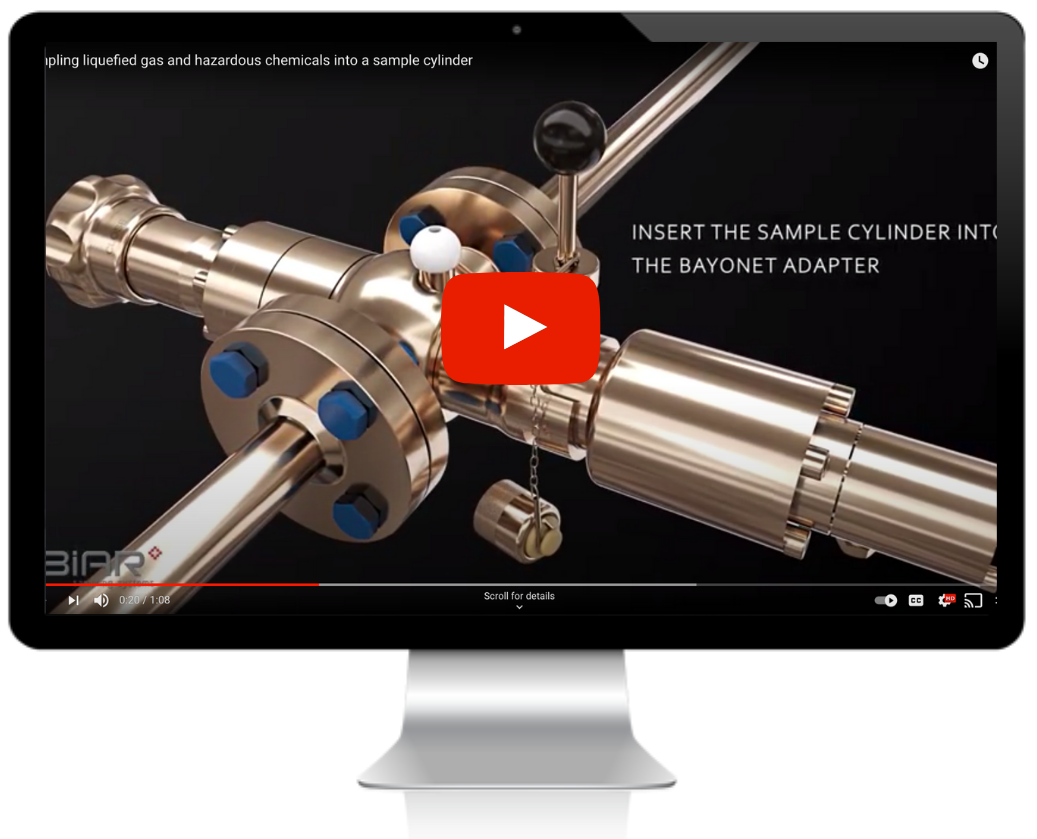
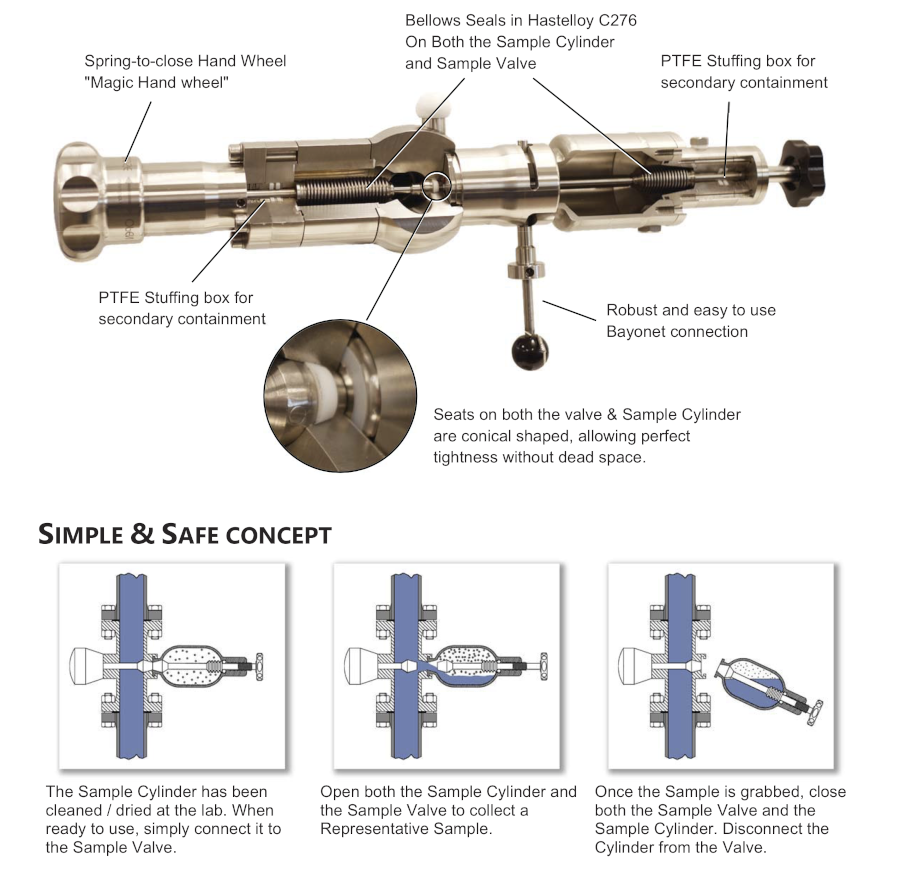
Specific Hazards Related to Sampling: Chlorine is an extremely hazardous and reactive chemical. Sampling must be carried out in sealed containers to reduce exposure risk and minimize releases into the environment. Chlorine will react with moisture, corroding metals such as carbon and stainless steel.
Alloys such as Hastelloy® or Monel are best suited for chlorine sampling due to their high corrosion resistance. Systems should also be designed so that no moisture can be introduced to influence the sample (e.g., condensation on the outside of fittings).
When sampling chlorine, allowance must also be made for thermal expansion within the receptacle. Ideally, there should be sufficient space within the container to prevent the danger of over-expansion.
![]()
3) Sampling Facts
a. Why plants need to sample
Plants need to sample chlorine to test its purity.
b. What are they looking / measuring in the sample
Non-volatiles and iron: Liquid chlorine is evaporated in a flask and the remains are measured.
Inerts: Liquid chlorine is put through a gasifier which turns it to gas, then it is run through a Gas Chromatograph (GC).
Other samples can be run using an Inductively Coupled Plasma Mass Spectrometer (ICP-MS) to test for organics.
4) Most common Sample Receptacles
Chlorine liquefied gas should be sampled into a closed container or cylinder. The receptacle should have sufficient volumetric space to allow for product expansion